 Section 2: Information, neurons, and spikes Tutorial 3: Simultaneous fitting/regressionĮxample Model Project: the Train Illusion 
Tutorial 4: Model-Based Reinforcement Learning Tutorial 2: Learning to Act: Multi-Armed Bandits Tutorial 2: Optimal Control for Continuous State Tutorial 1: Optimal Control for Discrete States Tutorial 1: Sequential Probability Ratio Testīonus Tutorial 4: The Kalman Filter, part 2īonus Tutorial 5: Expectation Maximization for spiking neurons Tutorial 2: Bayesian inference and decisions with continuous hidden state Tutorial 1: Bayes with a binary hidden state Tutorial 3: Synaptic transmission - Models of static and dynamic synapsesīonus Tutorial: Spike-timing dependent plasticity (STDP)īonus Tutorial: Extending the Wilson-Cowan Model Tutorial 1: The Leaky Integrate-and-Fire (LIF) Neuron Model Tutorial 3: Combining determinism and stochasticity Tutorial 3: Building and Evaluating Normative Encoding Modelsīonus Tutorial: Diving Deeper into Decoding & Encoding 
Tutorial 2: Convolutional Neural Networks Tutorial 4: Nonlinear Dimensionality Reduction Tutorial 3: Dimensionality Reduction & Reconstruction Tutorial 6: Model Selection: Cross-validation Tutorial 5: Model Selection: Bias-variance trade-off Tutorial 4: Multiple linear regression and polynomial regression Tutorial 3: Confidence intervals and bootstrapping Tutorial 1: Differentiation and Integration There might be decreases in freedom in the rest of the universe, but the sum of the increase and decrease must result in a net increase.Prerequisites and preparatory materials for NMA Computational Neuroscienceīonus Tutorial: Discrete Dynamical Systems The freedom in that part of the universe may increase with no change in the freedom of the rest of the universe. Statistical Entropy - Mass, Energy, and Freedom The energy or the mass of a part of the universe may increase or decrease, but only if there is a corresponding decrease or increase somewhere else in the universe.Qualitatively, entropy is simply a measure how much the energy of atoms and molecules become more spread out in a process and can be defined in terms of statistical probabilities of a system or in terms of the other thermodynamic quantities. 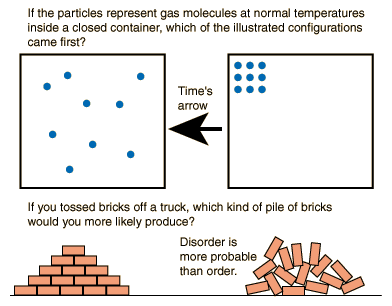
Statistical Entropy Entropy is a state function that is often erroneously referred to as the 'state of disorder' of a system.Phase Change, gas expansions, dilution, colligative properties and osmosis. Simple Entropy Changes - Examples Several Examples are given to demonstrate how the statistical definition of entropy and the 2nd law can be applied.A microstate is one of the huge number of different accessible arrangements of the molecules' motional energy* for a particular macrostate. Instead, they are two very different ways of looking at a system. Microstates Dictionaries define “macro” as large and “micro” as very small but a macrostate and a microstate in thermodynamics aren't just definitions of big and little sizes of chemical systems.“Disorder” was the consequence, to Boltzmann, of an initial “order” not - as is obvious today - of what can only be called a “prior, lesser but still humanly-unimaginable, large number of accessible microstate it was his surprisingly simplistic conclusion: if the final state is random, the initial system must have been the opposite, i.e., ordered. ‘Disorder’ in Thermodynamic Entropy Boltzmann’s sense of “increased randomness” as a criterion of the final equilibrium state for a system compared to initial conditions was not wrong. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
